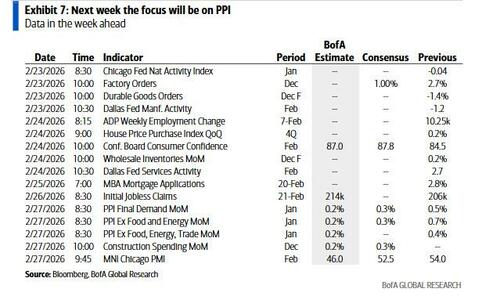
“Worst-Case Scenario”: Novo Nordisk Plunges After Next-Gen Obesity Drug Falls Short Of Lilly Rival
Shares of Novo Nordisk A/S plummeted again on Monday after the company reported trial results showing its next-generation obesity shot, CagriSema, delivered 20.2% weight loss at 84 weeks, compared to 23.6% for Eli Lilly & Co.’s tirzepatide (Zepbound).
Bloomberg Intelligence analyst Michael Shah explained, “This outcome is the worst-case scenario for Novo and heightens the need for M&A with Novo’s other GLP-1/Amylin drug.”
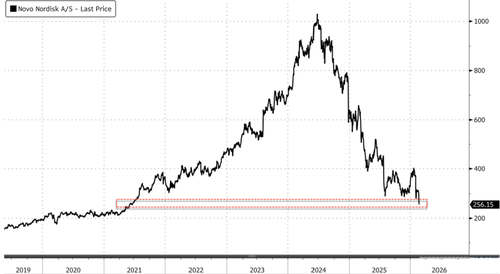
Shares of Novo in Copenhagen plunged as much as 16.5%, breaking below a support level that had held since August 2025. The stock is now down about 75% from its 2024 peak and is near its 2021 low.
The result is yet more trouble for Novo’s new leadership, led by Mike Doustdar, following Lars Fruergaard Jørgensen’s recent exit, along with board turnover linked to disagreements over a turnaround plan to regain GLP-1 market share. There’s also the copycat GLP-1 compounding issue involving the telehealth firm Hims & Hers.
Novo’s strategy revolves around CagriSema as Wegovy and Ozempic face longer-term patent pressure; it combines semaglutide and another gut hormone called amylin. Early studies have shown mixed results, and at least one large trial failed to meet Novo’s targeted weight loss.
Another CagriSema trial, due later this year, could yield better results because it aims to move patients to the highest dose.
“Clearly this weakens Novo Nordisk’s competitive stance in the obesity market – especially if the obesity market develops into a ‘winner takes it all’ market,” Danske Bank Credit Research analyst Brian Borsting wrote in a note. “That said, we continue to believe that Novo Nordisk’s product portfolio in the obesity market is diversified and solid although we see today’s news as credit negative.”
Meanwhile, Goldman analyst and Novo super bull James Quigley provided clients with an update on the CagriSema trial:
This morning (23rd February), Novo announced that CagriSema did not achieve the primary endpoint of non-inferiority in REDEFINE-4, with weight loss of 23% for the CagriSema arm vs. 25.5% for the tirzepatide 15mg arm, after 84 weeks of treatment. Previously, we had said that in a non-inferiority scenario, taking the PoS of CagriSema down to 0% in obesity, leaving $5bn in sales only for cagrilintide monotherapy, would lead to a -12% impact to our DCF, all else equal, but noted that investor expectations were likely lower for CagriSema. These data points could further reduce market expectations for CagriSema, even ahead of the REDEFINE 11 trial (1H’27), and while we continue to expect approval for CagriSema and likely some use by physicians as part of a portfolio approach in obesity, investors are not likely to give credit here until the sales start to come though post approval. Novo is looking to explore higher doses of CagriSema with a Phase 3 trial planned for 2H26 – although we believe investors are unlikely to give any credit until the sales trajectory is seen. Therefore, given our expectations noted above, the share price reaction at the time of writing of c.-12% appears in line, as any residual potential for CagriSema moves out of the expectations built in for the stock. Continued momentum for the launch of the Wegovy pill is even more important, we believe, as shifting volumes to the oral market could be advantageous, given Novo has a more competitive profile on weight loss.
CagriSema failed to meet the primary endpoint of demonstrating non-inferiority vs tirzepatide on weight loss at 84 weeks. On an efficacy-estimand basis, 2.4mg/2.4mg CagriSema showed -23.0% weight loss at 84 weeks, vs 25.5% weight loss with 15mg tirzepatide over the same time period. On a treatment-regimen estimand basis, CagriSema showed -20.2% weight loss at 84 weeks vs -23.6% with 15mg tirzepatide. As a result, REDEFINE-4’s primary endpoint of CagriSema demonstrating non-inferiority on weight loss vs tirzepatide was not reached.
CagriSema showed a well-tolerated safety profile, per Novo. While no tolerability data was given, in the release Novo said that overall CagriSema appeared to show a well-tolerated and safe profile, with the most common AEs being GI AEs. Of these, the vast majority were mild to moderate and improved over time, which was consistent with other drugs in the GLP1 agonist class.
In terms of next steps, Novo anticipates a decision from the FDA on CagriSema by y/e 2026. This is following the company’s submission to the FDA in December 2025 based on data from REDEFINE 1 and 2. Novo also expects to initiate an additional Phase 3 trial of higher dose CagriSema in 2H26, and expects the readout from REDEFINE 11 of 2.4mg/2.4mg CagriSema in 1H27 (longer term trial looking at the full weight loss potential of CagriSema in obesity).
Not surprisingly, Quigley’s continued “Buy” rating on the stock even as it has collapsed 75% from the peak and nears 2021 lows. “We are Buy rated on Novo Nordisk,” he said.
Related:
Novo Nordisk CEO To Step Down Following Brutal Bear Market
“Big Miss”: Wall Street Disappointed After Dismal Novo Nordisk GLP-1 Sales Outlook, Shares Plunge
GLP-1 Feud: HIMS Fires Back At Novo Nordisk, Slams Lawsuit As “Blatant Attack” By Big Pharma
GLP-1 Anti-Obesity U.S. Drug Market In Four Charts
Bloomberg data shows 17 “Buy” ratings, 14 “Hold” ratings, and 3 “Sell” ratings on Novo. The average 12-month price target among Wall Street analysts is 353 kroner.
How many Goldman clients are furious with Quigley’s Novo coverage?
Tyler Durden
Mon, 02/23/2026 – 09:25